What is covered under IVDR?
Any in vitro diagnostic medical device that is a reagent, reagent product, calibrator, control material, kit, instrument, apparatus, piece of equipment, software or accessory, whether used alone or in combination, intended by the manufacturer to be used in vitro for the examination of specimens, including blood and tissue donations, derived from the human body, solely or principally to provide information on one or more of the following:
- Concerning a physiological or pathological process or state
- Concerning congenital physical or mental impairments
- Concerning the predisposition to a medical condition or a disease
- To determine the safety and compatibility with potential recipients
- To predict treatment response or reactions
- To define or monitoring therapeutic measures
Specimen receptacles shall also be deemed to be in vitro diagnostic medical devices.
What is the difference between IVDR and IVDD?
Some of the main differences between the new In Vitro Diagnostic Regulation and the previous In Vitro Diagnostics Directive are: 1,2
- Scope and classification of products
- Improved transparency and data for the European Database for Medical Devices (EUDAMED), the European Union’s database for medical devices
- Role of Economic Operators (clarification of obligations of manufacturers, authorized representatives, importers and distributors)
- Changes to notified bodies
- Addition of Unique Device Identification (UDI) System for every IVD device
- Changes to clinical evaluation processes with an increase in clinical evidence and conformity assessment
Device risk-classification categories
What is the IVDR implementation timeline?
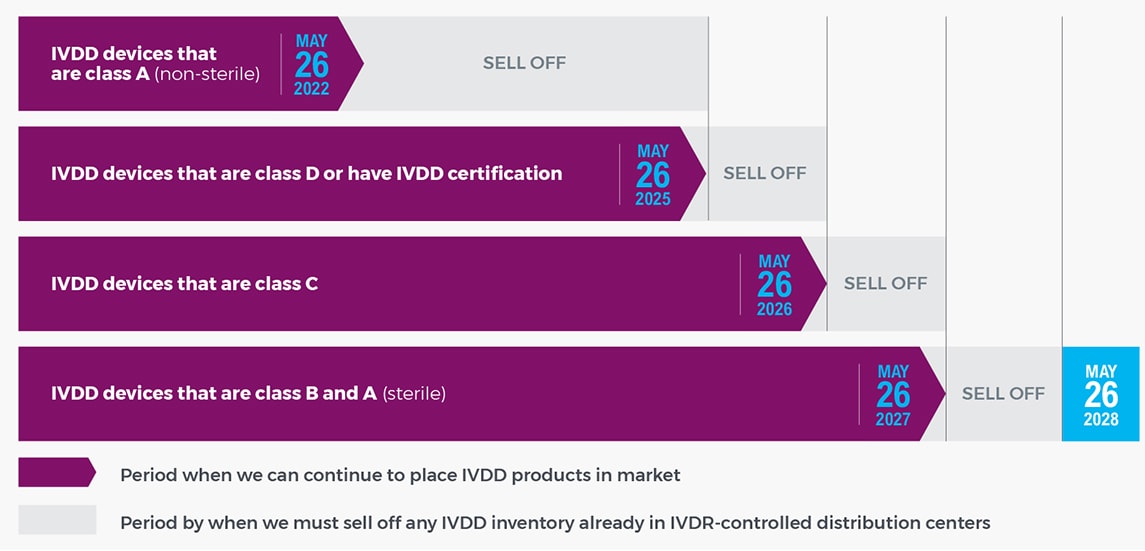
How does this impact me as a customer?
Beckman Coulter is working to ensure all relevant products are IVDR-compliant and have processes in place for recertification to ensure product continuity for all our customers.4
IFU Changes
IFUs will be updated to include the CE Notified Body Number alongside the CE mark.
Other changes to IFUs may include:
- Intended purpose statements
- Warnings or contraindications
- Additions or alterations to sample types, stability, interfering substances, linearity, reference ranges, specificity and sensitivity
- Manufacturer’s telephone number
- Updates to supporting references
Labeling changes
You can expect to see changes to our product labeling.
For vials, tubes and cartridges, you may see:
- Addition of IFU symbol and web address for IFUs
- Addition of phone number under BEC address
- Addition of target fill volume
- Removal of approximate concentration from vial labels
- Removal of hazard codes
For product boxes, you will see the addition of the IVDR-required notified body number next to the CE mark.
Beckman Coulter Diagnostics products that will be covered under IVDR
References:
*MD Buyline. User Satisfaction Trending for Beckman Coulter Microbiology Systems. October 1, 2016.
1. 6 Major Differences Between EU’s MDR/IVDR and MDD/IVDD. (2019, March 14). RegDesk. https://www.regdesk.co/major-differences-eu-mdr-mdd/
2. FAQS: IN VITRO DIAGNOSTIC MEDICAL DEVICE REGULATION (IVDR). (2021). TUV. https://www.tuvsud.com/en-us/industries/healthcare-and-medical-devices/medical-devices-and-ivd/medical-device-market-approval-and-certification/eu-in-vitro-diagnostic-medical-device-regulation/faqs-in-vitro-diagnostic-medical-device-regulation-ivdr
3. https://www.medtecheurope.org/new-medical-technology-regulations/transition-periods/ accessed May 10, 2022
4. https://www.medtecheurope.org/wp-content/uploads/2020/05/20200526_Impact_Changes_Int_Reg_invitrodiagnostics_IVDR.pdf
 English
English

