Efficiently support diagnosis and assessment of heart failure severity with trustworthy results
B-type natriuretic peptide (BNP) and the N-terminal pro hormone BNP (NT-proBNP) are well established for use in diagnosis of heart failure. Following production, the prohormone is cleaved at a 1:1 ratio into NT-proBNP and BNP. (Fig. 1) BNP is involved in regulating blood volume.2 Blood levels of NT-proBNP and BNP increase when the left ventricle is stretched, as occurs during heart failure, a condition where the heart cannot pump enough blood.
Natriuretic peptides play a significant role in controlling cardiovascular function. The biologically active BNP ring is involved in cardiorenal homeostasis by stimulating diuresis and natriuresis. Both NT-proBNP and BNP biomarkers provide demonstrated benefit for heart failure patients in emergency and outpatient settings. Both NT-proBNP and BNP are included in standard of care guidelines for both the American College of Cardiology/American Heart Association5 and European Society of Cardiology.6
Laboratories can help clinicians deliver patient care with confidence for all stages of heart failure with Access NT-proBNP and BNP assays, highly sensitive and specific assays that aid clinicians in heart failure diagnosis, assessment of severity, and risk stratification.
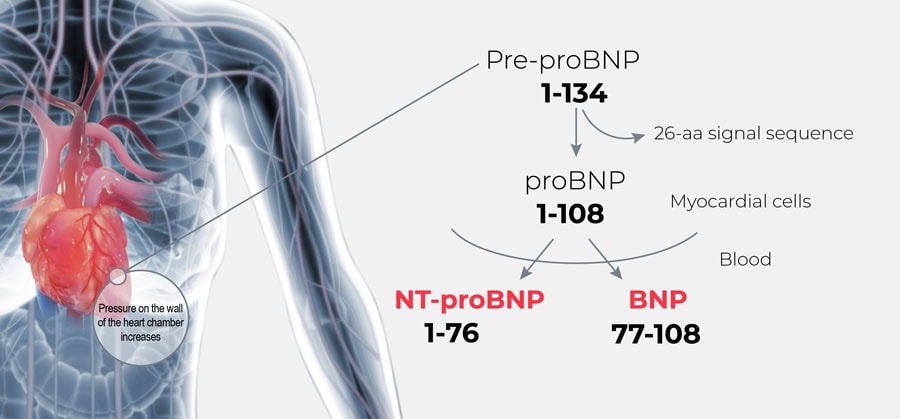
Figure 1. BNP and NT-proBNP production in the ventricular cardiac myocardium7
 English
English























