Access AMH advantages over other current AMH immunoassays
- Increase physician and patient confidence with a recombinant human AMH calibrator that is more stable and accurately mirrors AMH in patient samples for greater immunological recognition of antibodies*
- Experience a 16 times increase in sensitivity
One test, multiple uses
Diagnostic testing of AMH levels is used in:
- Fertility assessments. AMH testing has significant advantages over other tests, including increased sensitivity and specificity, and consistency in serum levels throughout the menstrual cycle (see Figure 1)1,2
Figure 1. Mean AMH Values across Menstrual Cycle1
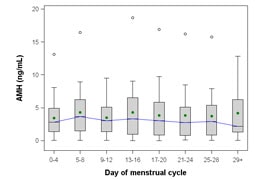
AMH levels can be reliably measured during any day of the menstrual cycle.
- Reproductive aging evaluations. AMH levels decline earlier than other signs of menopause—such as increasing follicle-stimulating hormone (FSH) levels and irregular menses—and could be used to identify age-related infertility3,4
Access AMH advantages over manual assays
- Reduce testing costs by 16 percent by using an automated AMH test*
- Decrease technologist handling time by 80 percent and allow for greater walk-away time through random access capabilities and once-a-month system calibration*
- Achieve 4 times quicker TAT—40 minutes with an automated assay compared with 170 minutes with ELISA*
Evidence for the Access AMH assay
- The synthetic Access AMH matrix [simulates analyte-free human serum]…[and] is expected to ensure batch-to-batch consistency and assay performance and minimize detection limitations5
- The automated assay exhibits high levels of stability and sensitivity and shows correlation with the existing ELISA method and between analyzer platforms6
Discover a full line of reproductive assays to meet your testing needs
Perform a broad range of reproductive testing services with our comprehensive assay portfolio:
- AFP (ONTD)
- AMH
- DHEA-S
- hFSH
- hLH
- Inhibin A†
- Inhibin B†
- PAPP-A
- Progesterone
- Prolactin
- Sensitive Estradiol
- SHBG
- Testosterone
- Total βhCG (5th IS)
- Unconjugated Estriol
Access AMH is exclusively available for Beckman Coulter Access and DxI immunoassay analyzers. Access AMH setup is quick and easy.
 English
English






